First Woman with Neuralink Chip Achieves Thought-Controlled Handwriting
Audrey Crews Makes Neuralink History: Writes Name with Thought-Controlled Brain Implant
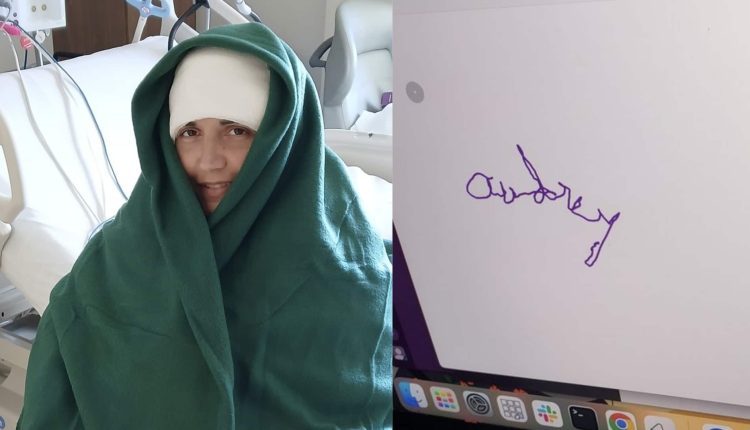
In a defining moment for neurotechnology, Audrey Crews has become the first woman to successfully scribble her name on a laptop screen using only her thoughts and a Neuralink brain-computer interface (BCI). The achievement demonstrates significant progress in converting neural signals into digital actions, offering new pathways to digital independence for people with severe paralysis.

How Neuralink’s Technology Works
Crews received Neuralink’s N1 Implant, a device the size of a coin, surgically placed in her motor cortex by the company’s R1 Robot. The implant contains 1,024 electrodes distributed across ultra-fine threads thinner than human hair, designed to detect electrical signals generated when she imagines moving her hand. These signals are wirelessly transmitted to a computer running Neuralink’s decoding software, which translates her intention into cursor movements and clicks. While earlier Neuralink patients like Nolan Arbaugh showcased gaming and web browsing, Crews’ precise handwriting marks a leap in dexterity and calibration accuracy.
Overcoming Technical and Biological Hurdles
The milestone follows significant challenges. Previous Neuralink participants experienced electrode “retraction” and signal degradation, requiring software patches to maintain functionality. Crews’ success suggests Neuralink may be improving system stability. Still, users face daily recalibration, a 45-minute process where patients “retrain” the device by visualizing movements. Dr. Anya Sharma, a neuroengineer unaffiliated with Neuralink, notes: “Handwriting requires extraordinarily fine motor signal decoding. If sustained, this precision could accelerate real-world adoption for communication.”
Global Expansion Amid Rigorous Screening
Neuralink is scaling its trials internationally. Following U.S. and Canadian studies, Great Britain’s UCLH hospital will lead the GB-PRIME trial, recruiting up to seven participants with paralysis or neurological conditions like ALS. Applicants must meet strict criteria: significant limb impairment or paralysis, age 18+, and residency in trial countries. Though thousands have joined Neuralink’s patient registry, only a fraction qualify. “We’re only accepting individuals with qualifying conditions,” the company states.
Competition and the Road Ahead
While Neuralink dominates headlines, rivals like Precision Neuroscience and Synchron advance less invasive alternatives. Precision’s surface-mount electrode film avoids brain penetration, potentially easing FDA approval. Meanwhile, Blackrock Neurotech, with decades of BCI research, enabled a paralyzed man to feel touch via a robotic arm in 2021. Neuralink aims to integrate robotic limb control in its CONVOY study soon.
A Cautiously Optimistic Future
Crews’ achievement underscores BCI’s potential to restore agency, yet significant barriers remain. Clinical trials will take approximately six years, and experts emphasize that commercial availability requires larger safety studies. Michael Mager, CEO of Precision Neuroscience, acknowledges the field’s momentum but tempers expectations: “We’re still solving data transmission, wireless power, and long-term biocompatibility”. For now, Crews’ signature symbolizes hope, a tangible step toward redefining life with paralysis.
Subscribe to my whatsapp channel

Comments are closed.